Improving Clinical Outcomes
Theraworx Protect’s line of products includes clinically shown, safe, topical solutions trusted by top hospitals across the country to help manage healthcare-associated risks.
Theraworx Protect stands as a proven and non-inferior alternative to traditional ICU bathing methods*. Embrace the convenience of an all-in-one solution, eliminating the necessity for additional surfactant-based hygiene items, saline, or barrier creams. Our innovative no-rinse, non-irritating formula ensures a refreshing cleanse without leaving any undesirable sticky residue behind. Elevate your skincare routine with a product designed for effectiveness and comfort.
*As compared to standard ICU bathing in relevant studies.

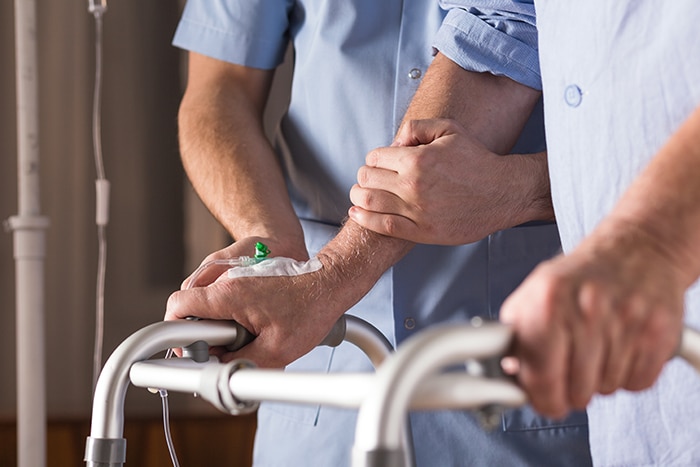
Adult Care Solutions
As part of an infection control bundle, Theraworx Protect has proven to be a key tool in helping to maintain the catheter and lower CAUTI/CLABSI rates in adults. Safe for use on compromised skin, our low-pH formulation supports the skin’s natural antimicrobial barrier and defensive functions which is why many hospitals have made the switch to Theraworx Protect.

Pediatric Care Solutions
As part of an infection control bundle, Theraworx Protect provides advanced total body and perineum care and helps to maintain the catheter and lower CAUTI/CLABSI rates in pediatric care units. It’s safe for use on compromised skin and has a low-pH formulation that supports the skin’s natural antimicrobial barrier and defensive functions.
Learn how to add Theraworx Protect to your pediatric bathing and infection control bundles.
Learn about the potential financial benefits of Theraworx Protect as a part of your bathing and infection control bundles.
Learn how Theraworx Protect can help reduce hospital-acquired infections as a part of an infection control bundle.
Learn how to add Theraworx Protect to your adult bathing and infection control bundles.
Resources and CE
Get Theraworx product support information and details for how to use.
You can also browse our CE resources, see application videos, and see our Safety Data.


Contact Us
Check with your healthcare facility’s GPO or distribution partner to order Theraworx Protect. Or, contact us to connect with your local Theraworx Protect representative about ordering, or to request specific product protocols.



